- Home
- CBSE
- ICSE
- NCERT
- Text Book Solutions
- Our Products
- More
Redox reaction is related to gain or loss of electrons. Reaction in which oxidation and reduction takes place simultaneously is called redox reaction. This chapter deals with problems based on redox reactions, oxidation number and balancing of redox reactions by ion, electron method and oxidation number method.
Oxidation is defined as the addition of oxygen/electronegative element to a substance or rememoval of hydrogen/ electropositive element from a susbtance.
2Mg(s) + O2(g) ⟶ 2MgO(s)
Mg(s) + Cl2(g) ⟶ MgCl2(s)
Reduction is defined as the memoval of oxygen/electronegative element from a substance or addition of hydrogen or electropositive element to a substance.
2FeCl3(aq) + H2(g) ⟶ 2FeCl2(aq) + 2HCl(aq)
2HgO(s) ⟶ 2Hg(l) + O2(g)
Reaction in which oxidation and reduction takes place simultaneously is called redox reaction. Oxidation and reduction are complementary to each other, one cannot take place alone. So both oxidation and reduction will occur simultaneously. It is obvious that if a substance takes electrons there must be another substance to give up these electrons.
2FeCl3 + SnCl2 ⟶ 2Fecl2 + SnCl4
Oxidation number for an element is the arbitrary charge present on one atom when all other atoms bonded to it are removed. For example, if we consider a molecule of HCl, the Cl atom is more electronegative than H-atom, therefore, the bonded electrons will go with more electronegative chlorine atom resulting in formation of H+ and Cl– ions. So oxidation number of H and Cl in HCl are +1 and –1 respectively.
The following points are important to determine the oxidation number of an element.
A substance which undergoes oxidation acts as a reducing agent while a substance which undergoes reduction acts as an oxidising agent. For example, we take a redox reaction,
Zn + Cu2+ ⟶ Zn2+ + Cu
In this reaction, Zn is oxidised to Zn2+ so Zn is reducing agent and Cu2+ is reduced to Cu so Cu2+ is an oxidising agent.
A combination reaction is a reaction in which two or more substances combine to form a single new substance. Combination reactions can also be called synthesis reactions. The general form of a combination reaction is:
A + B ⟶ AB
Na(s) + Cl2(g) ⟶ 2NaCl(s)
A decomposition reaction is a reaction in which a compound breaks down into two or more simpler substances. The general form of a decomposition reaction is:
AB ⟶ A + B
2HgO(s) ⟶ 2Hg(l) + O2(g)
Displacement reaction is a chemical reaction in which a more reactive element displaces a less reactive element from its compound.
CuSO4(aq) + Zn(s) ⟶ ZnSO4(aq) + Cu
The reactions in which a single reactant is oxidized and reduced is known as Disproportionation reactions. The disproportionation reaction is given below
2H2O2 ⟶ 2H2O + O2
In this method number of electrons lost in oxidation must be equal to number of electrons gained in reduction. Following rules are followed for balancing of reactions:

A galvanic cell or voltaic cell is simple electrochemical cell in which a redox reaction is used to convert chemical energy into electrical energy. It means electricity can be generated with the help of redox reaction in which oxidation and reduction takes place in two separate compartments. Each compartment consists of a metallic conductor and dipped in suitable electrolytic solution of same metal. Metallic rod acts as electrode.
The compartment having electrode dipped in solution of electrolyte is known as half cell and a half cell has a redox couple. A redox couple means a solution having reduced and oxidised form of a substance together, taking part in oxidation or reduction half reaction. It is depicted as M+n / M i.e., oxidised form / reduced form. To prepare a galvanic cell two half cells are externally connected through a conducting wire and internally through salt bridge.
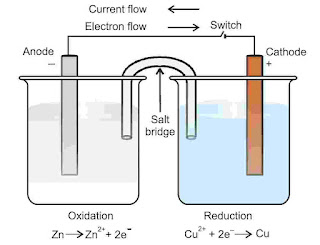
Anodic oxidation : Zn2 ⟶ Zn+2(aq) + 2e(s)
Cathodic reduction : Cu+2(aq) + 2e ⟶ Cu(s)
Net reaction : Zn(s) + Cu+2(aq) ⟶ Zn+2(aq) + Cu(s)
This cell can be briefly presented in one line, known as cell notation i.e.,
Zn | Zn+2 || Cu+2 | Cu